|
8/10/2023 0 Comments Electron capture chemistry 
Species diversity generally decreases with increasing elevation. There are many factors correlated with this gradient that may affect species diversity, such as average temperature and precipitation, the variability in temperature and precipitation, annual net primary productivity, and geological history. Diversity is highest in the tropics and lowest at the poles. 
Latitude, altitude, productivity, spatial heterogeneity, size, age, and disturbance regime.įor most groups of terrestrial plants and animals, diversity decreases with increasing latitude. Thus, the broadest classification level for organisms is domain, which includes Archaea, Bacteria, and Eukarya. 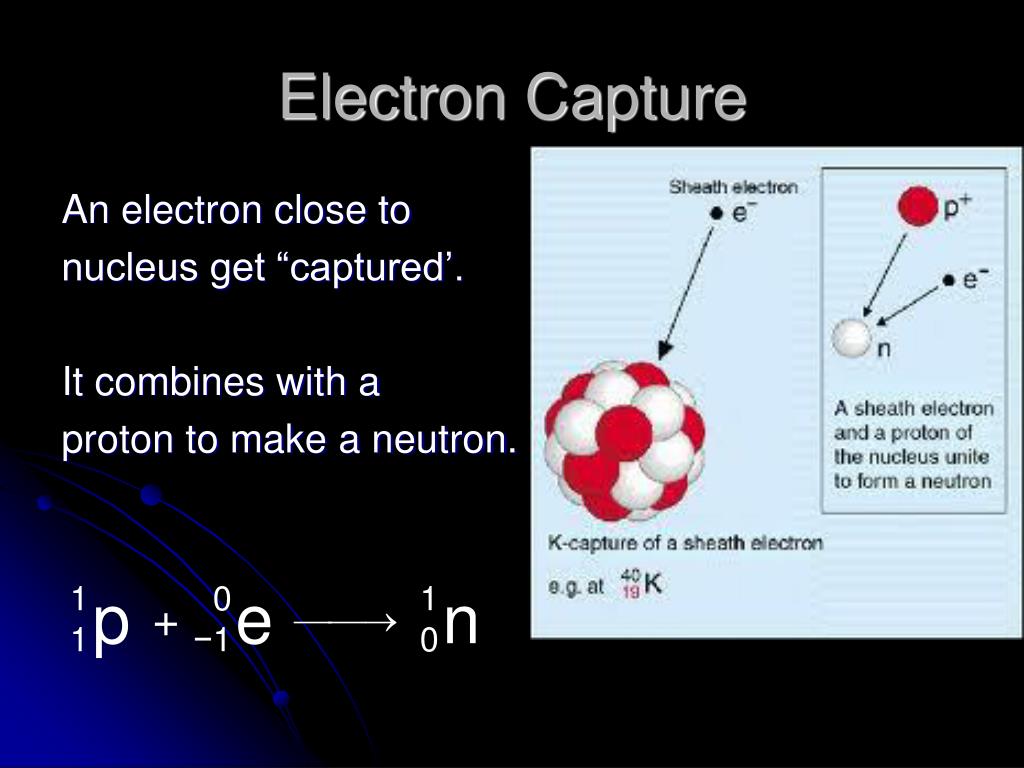
The Archaea and bacteria are all prokaryotes, single-celled microorganisms without a membrane-bound nucleus, while eukarya includes all organisms that have a cell nucleus and other membrane-bound organelles. From broadest to most specific they include: Domain, Kingdom, Phylum, Class, Order, Family, Genus and species while the Domain is the broadest category which included the most individual organisms.Īccording to the domain system, the tree of life consists of either three domains that are Archaea, Bacteria and Eukarya or two domains consisting of Archaea and Bacteria, with Eukarya included in Archaea. The classification is made up of Eight broad categories. The broadest classification level for organisms is domain, which includes Archaea, Bacteria, and Eukarya. It is clear that for an isotope, the number of electrons and neutrons will vary but the number of protons will always be fixed. That is why we cannot consider electrons to be always same for isotopes of an element. Also, in a neutral atom number of protons is equal to number of electrons but when an atom becomes negatively or positively charged, it can gain or loose electrons so electrons have tendency to vary. During chemical reactions, it is electron which directly participates, while proton and neutrons do not take part or move during chemical reactions. Electrons are extremely light in weight so they do not contribute towards weight but they have a great role in chemical reactions. Apart from that, an atom has electrons too but they are outside the nucleus. As we know that every atom has a nucleus which has protons and neutrons and due to these protons and neutrons an atom has weight. If number of neutrons of an atom vary but number of protons in them are same then it simply means that they both are isotopes of a particular element. If two atoms are isotopes of the same element then the number of protons in those two atoms must be same but the number of neutrons must not be same. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
